Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Aritomo, Yoshihiro*; Iwamoto, Akira*; Nishio, Katsuhisa; Ota, Masahisa*
Physical Review C, 105(3), p.034604_1 - 034604_8, 2022/03
Times Cited Count:2 Percentile:52.69(Physics, Nuclear)Aritomo, Yoshihiro*; Miyamoto, Yuya*; Nishio, Katsuhisa
Nihon Butsuri Gakkai-Shi, 75(10), p.631 - 636, 2020/10
no abstracts in English
 decay of
decay of  Fm
Fm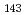 and
and 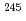 Fm
Fm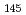 , and of their daughter nuclei
, and of their daughter nucleiKhuyagbaatar, J.*; He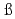 berger, F. P.*; Hofmann, S.*; Ackermann, D.*; Burkhard, H. G.*; Heinz, S.*; Kindler, B.*; Kojouharov, I.*; Lommel, B.*; Mann, R.*; et al.
berger, F. P.*; Hofmann, S.*; Ackermann, D.*; Burkhard, H. G.*; Heinz, S.*; Kindler, B.*; Kojouharov, I.*; Lommel, B.*; Mann, R.*; et al.
Physical Review C, 102(4), p.044312_1 - 044312_8, 2020/10
Times Cited Count:8 Percentile:69.78(Physics, Nuclear)Miyamoto, Yuya*; Aritomo, Yoshihiro*; Tanaka, Shoya; Hirose, Kentaro; Nishio, Katsuhisa
Physical Review C, 99(5), p.051601_1 - 051601_7, 2019/05
Times Cited Count:16 Percentile:84.48(Physics, Nuclear)Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:27 Percentile:69.46(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Iwamoto, Akira; M ller, P.*; Madland, D. G.*; Sierk, A.*
ller, P.*; Madland, D. G.*; Sierk, A.*
Journal of Nuclear Science and Technology, 39(4), p.332 - 336, 2002/04
Times Cited Count:2 Percentile:17.03(Nuclear Science & Technology)The theoretical calculations on the most probable mass division in nuclear fission process are given. The model is based on the topographical analysis of the five-dimensional potential energy surface of more than 2.5 million mesh points. Special attention is paid to obtain the energies and deformations of the saddle points without using any approximation. The calculation tells us that there exist multiple saddle points in the static potential energy surface. The lowest and the next lowest saddle point play an important role for the fission process. Of these two saddle points, one is mass-symmetrically deformed and the other is mass-asymmetrically deformed and the relative heights of these two saddle points depend on the fissioning nuclei. In case of Fm isotopes, the relative height of two saddle points appears very sensitively, i.e.,  Fm has asymmetrically deformed lowest saddle point and in
Fm has asymmetrically deformed lowest saddle point and in 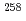 Fm, the lowest saddle point is symmetrically deformed. This feature explains the phenomenon called bimodal fission.
Fm, the lowest saddle point is symmetrically deformed. This feature explains the phenomenon called bimodal fission.
Sato, Tetsuya; Asai, Masato; Kaneya, Yusuke*; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Takeda, Shinsaku*; Vascon, A.*; Sakama, Minoru*; Sato, Daisuke*; et al.
no journal, ,
The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Sato, Tetsuya
no journal, ,
The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. In the present study, we have successfully determined IP
of lawrencium (Lr, Z = 103) using a surface ionization method. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Sato, Tetsuya; Asai, Masato; Kaneya, Yusuke*; Tsukada, Kazuaki; Toyoshima, Atsushi; Mitsukai, Akina*; Takeda, Shinsaku*; Vascon, A.*; Sakama, Minoru*; Sato, Daisuke*; et al.
no journal, ,
The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Sato, Tetsuya
no journal, ,
The first ionization potential (IP ), a measure of the energy required to remove one valence electron from a neutral atom, yields information on the valence electronic structure. Recently, we successfully determined the IP
), a measure of the energy required to remove one valence electron from a neutral atom, yields information on the valence electronic structure. Recently, we successfully determined the IP value of the heaviest actinide element, lawrencium (Lr,
value of the heaviest actinide element, lawrencium (Lr, 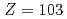 ) for the first time in an atom-at-a-time scale using a surface ionization method. The experimental result has shown that the IP
) for the first time in an atom-at-a-time scale using a surface ionization method. The experimental result has shown that the IP of Lr is distinctly low among actinide elements owing to the configuration of closed
of Lr is distinctly low among actinide elements owing to the configuration of closed 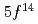 and
and 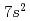 shells with an additional weakly-bound electron in the valence orbital. As a next step, we have applied this method to IP
shells with an additional weakly-bound electron in the valence orbital. As a next step, we have applied this method to IP measurements of other heavy actinide elements, No, mendelevium (Md,
measurements of other heavy actinide elements, No, mendelevium (Md, 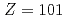 ) and fermium (Fm,
) and fermium (Fm, 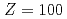 ). The experimental results of the three elements are in good agreement with the predicted ones obtained by theoretical and/or semi-empirical calculations. The IP
). The experimental results of the three elements are in good agreement with the predicted ones obtained by theoretical and/or semi-empirical calculations. The IP value increased with an atomic number up to No and fell dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. This behavior clearly indicates that the
value increased with an atomic number up to No and fell dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. This behavior clearly indicates that the 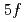 orbital is filled up at No.
orbital is filled up at No.